Chapter 4 Nuclear Fission
4.1 Introduction
We have seen (lecture 2) that the binding energy per nucleon reaches a maximum for . For heavier elements, it is therefore possible to achieve an increase in binding energy by splitting into smaller elements in a process known as nuclear fission. The fragments are more stable than the parent and the excess energy is released.
Fission can occur spontaneously but is not usually an important decay process for . For example, for decay of years, while for spontaneous fission of years.
It can also be induced, and this is how it was discovered by Hahn and Strassmann in 1938 when they were firing neutrons at heavy element targets to try to create radioactive elements. Instead, the heavy elements were observed to ‘split in half’. Other particles can be used to induce fission. For example, thorium undergoes fission with
-
•
1 MeV neutrons
-
•
9 MeV deuterons
-
•
32 MeV particles
-
•
7 MeV protons
The incident particle causes the nucleus to be distorted when it is absorbed. If the deformation is large enough the electrostatic repulsion between protons can overcome the strong force and the nucleus splits into two.
We shall be mainly interested in the fission of uranium. Its fission fragments are not identical and have masses spanning the range to . An example is
| (4.1) |
Note that the fission process itself generates neutrons – on average 2.44/fission reaction – and these neutrons can themselves induce new fission events. The ensuing chain reaction can occur rapidly as in a nuclear bomb, or under careful control in a fission reactor.
4.2 Fission energetics
4.2.1 Electrostatic viewpoint
We can regard the fission fragments of Eqn. 4.1 as forming within the parent nucleus and being in transient contact before fission occurs. This is similar to our treatment of decay, where the particle is pre-formed inside the nucleus. The electrostatic energy is then converted into kinetic energy as the fragments fly apart.
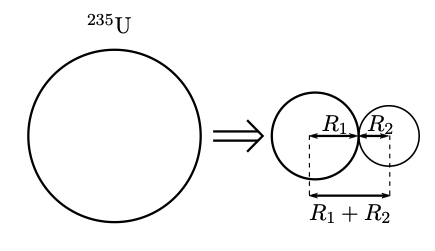
This does not happen instantaneously as there is a potential energy barrier to be overcome. Tunneling does not occur as readily as it does in decay because the barrier is wider (witness the very long half-life for the spontaneous fission of 238U). Rather, the activation energy is supplied by the incident particle.
Definition 4.2.1 (Activation Energy).
Amount of energy required to overcome the Coulomb potential energy barrier.
Example 4.2.1 (Energy Released from Fission).
Question: For the nuclear decay given by Eqn. 4.1 calculate the energy released and the height of the Coulomb barrier. The atomic masses are , and . Sketch the resulting effective nuclear potential.
Solution.
The energy released is then 180 MeV. The Coulomb barrier is found to be
where one can make use of the relation
Note the energy release of the transient states in the intermediate system is less than the Coulomb barrier. This means a more realistic potential barrier to fission is shown in Fig. 4.3.
4.2.2 Liquid drop model
An incident nucleon will also deform the nucleus. This can be modelled using the liquid drop model.
If we stretch an initially spherical nucleus, it may return to its spherical shape or, if sufficient energy is provided, it may split into two drops (Fig. 4.4).
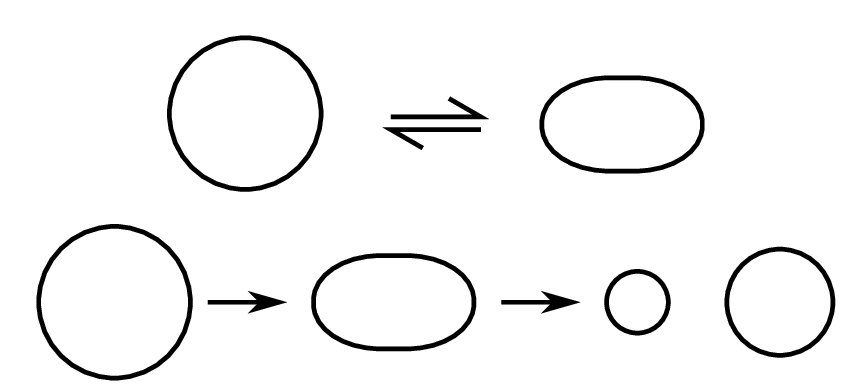
We can use the semi-empirical binding energy formula to see under what conditions this might occur. Only the surface and Coulomb terms depend on the shape of the nucleus. The volume, asymmetry and pairing terms are unaffected by the deformation, as the composition of the nucleus (i.e. the number of protons and neutrons) is unchanged.
Consider a nucleus deformed into a prolate ellipsoid, with a small deformation parameter , minor axis and major axis . The volume is constant between a sphere () and an ellipsoid (). One can show the surface area of the ellipsoid for small deformations is
| (4.2) |
The surface term of the liquid drop model is then modified to
| (4.3) |
The Coulomb term is modified to
| (4.4) | ||||
We can see how these terms change with eccentricity by differentiating with respect to :
| (4.5) | |||||
So, as increases the surface term leads to a decrease in binding energy, whereas the Coulomb term results in an increase. The condition for an overall increase is thus:
| (4.6) | |||||
If the deformed nucleus is more stable than the spherical nucleus and a positive feedback loop develops leading to fission. Note this simplified calculation must be modified for QM tunnelling, but in general serves as a reasonably good indicator of how likely a nucleus is to fission spontaneously.
If we take 238U as a typical heavy nucleus, . So, according to Eqn. 4.6 a nucleus for which would be unstable. This requires and hence , well beyond the mass of the known elements.
4.3 Power from nuclear fission
4.3.1 Fission cross section for 235U and 238U.
We have seen that incident particles (usually neutrons) are needed to induce fission. The probability of fission occurring for incident neutrons with a given energy is encoded in the cross-section. We will explore the relation between probability and cross-section later on, but a larger cross-section means a higher probability for fission. The cross-section has units of area, and in particle and nuclear physics is usually measured in barns, where .
Definition 4.3.1 (Barn).
The barn has units of area, where .
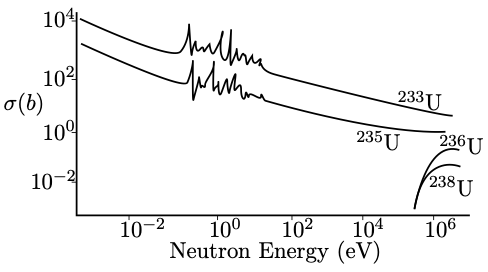
Fig. 4.5 illustrates the cross-sections for neutron-induced fission of uranium isotopes. Notice that 235U (and 233U) has a large reaction cross-section for neutrons in the thermal range but 238U (and 236U) requires neutrons of much higher energy. What is the explanation for this ‘even’ ‘odd’ effect?
First, we need to understand excitation energy:
Definition 4.3.2 (Excitation Energy).
Excitation is the addition of a discrete amount of energy (called excitation energy) to a nucleus that results in the lowest energy (or ground state) becoming one of higher energy (excited state). It is indicated by a .
The excitation energy when 235U captures a neutron to form the compound state is 6.5 MeV (see the worked example). The activation energy for 236U is 6.2 MeV. Therefore, when captures a neutron to form the excited state , the energy of the excited state exceeds the activation energy and is enough to induce fission even though the incident neutron has zero kinetic energy! The cross-section is therefore significant even for very low-energy neutrons.
A similar calculation for 238U gives a MeV, which is less than the activation energy of 239U (6.6 MeV). Therefore, when captures a neutron to form the excited state , the energy of the excited state is not enough to induce fission unless the incident neutron carries significant kinetic energy (of order MeV). The cross-section is therefore very small for low-energy neutrons.
Example 4.3.1 (Excitation Energy).
Calculate the excitation energy when 235U captures a neutron to form the compound state .
| (4.7) |
where the third line assumes the neutron’s kinetic energy is small.
The large difference in excitation energy can be understood from the pairing term of the semi-empirical binding energy formula, and is illustrated in Fig. 4.3.1. For example, the binding energy of 236U is increased by the pairing term (in other words the ground-state energy is lowered). The excitation energy is therefore increased over what it would be in the absence of pairing.
Difficult 4.3.1.
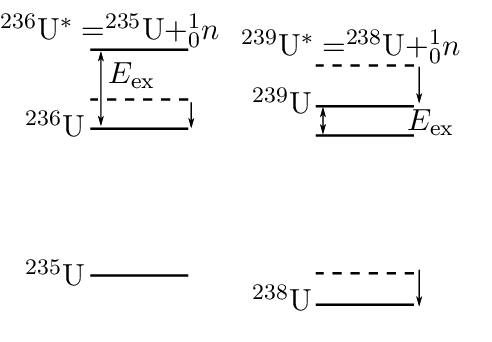
Effect of pairing on excitation energies. This diagram is a very common cause of confusion. The vertical axis is energy, with lower energy states nearer the bottom. The dashed lines indicate the ground state energy of 236U (left) and 238U (right) without including the pairing term. For these nuclei the pairing term increases the binding energy (they have even and even ), the nuclei are more stable and the ground-state energy is lowered. This is why the solid line is below the dashed line. Notice the opposite effects of including the pairing term on the excitation energies of 236U and 239U. For 236U it increases the excitation energy over what it would be in the absence of pairing, while for 239U it decreases the excitation energy over what it would be in the absence of pairing.
Example 4.3.2 (Pairing).
Question: Which of the following nuclei would you expect to have large cross-sections for fission induced by low-energy (thermal) nucleons. Use arguments based on the pairing energy. (i) (ii) (iii) (iv) (v) .
Solution.
(i)
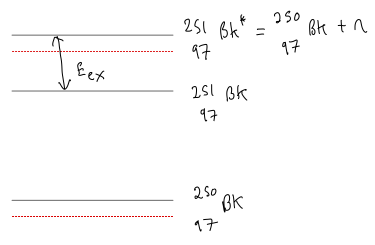
Increased so higher .
(ii)
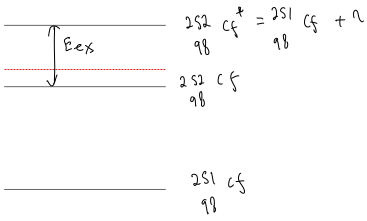
Increased so higher .
(iii)
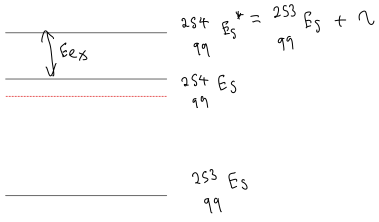
Decreased so lower .
(iv)
The energy level structure is the same as (ii).
Increased so higher .
(v)
The energy level structure is the same as (i).
Increased so higher .
4.4 Controlled fission
The primary fuel in nuclear reactors is 235U but natural uranium has an isotopic composition of 0.72% 235U, 99.28% 238U. It is possible to run reactors with natural uranium, but usually the fuel is enriched, typically to about 3%. Each fission event releases about 200 MeV (in the form of KE of the fragments and radiation). It also releases neutrons (both immediately, prompt neutrons, and after some delay, delayed neutrons) and it is these that sustain the chain reaction. The neutrons released are fast neutrons, for which the reaction cross-section is small (Fig. 4.5). It is advantageous to moderate these neutrons to thermal velocities where the reaction cross section is much higher. Light nuclei are the most effective moderators as the neutrons transfer more kinetic energy to them in elastic collisions. Fermi chose carbon and built the first chain reaction pile consisting of alternating layers of uranium and graphite in a squash court at the University of Chicago in 1942.
It is helpful to define a neutron reproduction factor that is the ratio of the number of neutrons in a given generation to the preceding one:
-
•
supercritical
-
•
critical
-
•
subcritical
To maintain a steady release of energy we would like the pile to be exactly critical. This depends on 4 principal factors – the four-factor formula.
-
1.
Even though each fission event produces on average 2.44 neutrons, not all of these will be available to induce subsequent fission events – some will be absorbed. The effective number is about 1.33 for natural uranium and 1.84 for 3% enriched uranium.
-
2.
These fast neutrons must be moderated and during this process, some will encounter 238U nuclei which have a small cross-section ( b) for fission by fast neutrons. This leads to a small increase in the number of neutrons which we account for by introducing the fast fission factor . For natural uranium has a value of about 1.03.
-
3.
As the fast neutrons are thermalized by collisions with the moderator (e.g. carbon) they pass through the eV energy range in which 238U has many capture resonances (not resulting in fission) with cross sections in the range of 1000 b. To thermalize the neutrons requires about 100 collision with carbon nuclei over a distance of about 19 cm. We can organise the nuclear pile as a matrix of uranium fuel rods, separated by about 19 cm of graphite to achieve this. But inevitability some neutrons will be captured by 238U during thermalization. We introduce the resonance escape probability to account for this. Typically might have a value of 0.9.
-
4.
One reason for choosing carbon as a moderator is that it has a very small probability for capture of thermal neutrons (cross-section = 0.0034 b), but there is inevitably a lot present, so we introduce the thermal utilization factor to give the fraction of thermal neutrons available to 235U and 238U. Again, this factor is typically around 0.9.
Putting these factors together we have the four-factor formula for the reproduction factor
| (4.8) |
4.4.1 Fission reactors
A common type of reactor used for power generation is the pressurised water reactor (PWR) illustrated in Fig. 4.6.
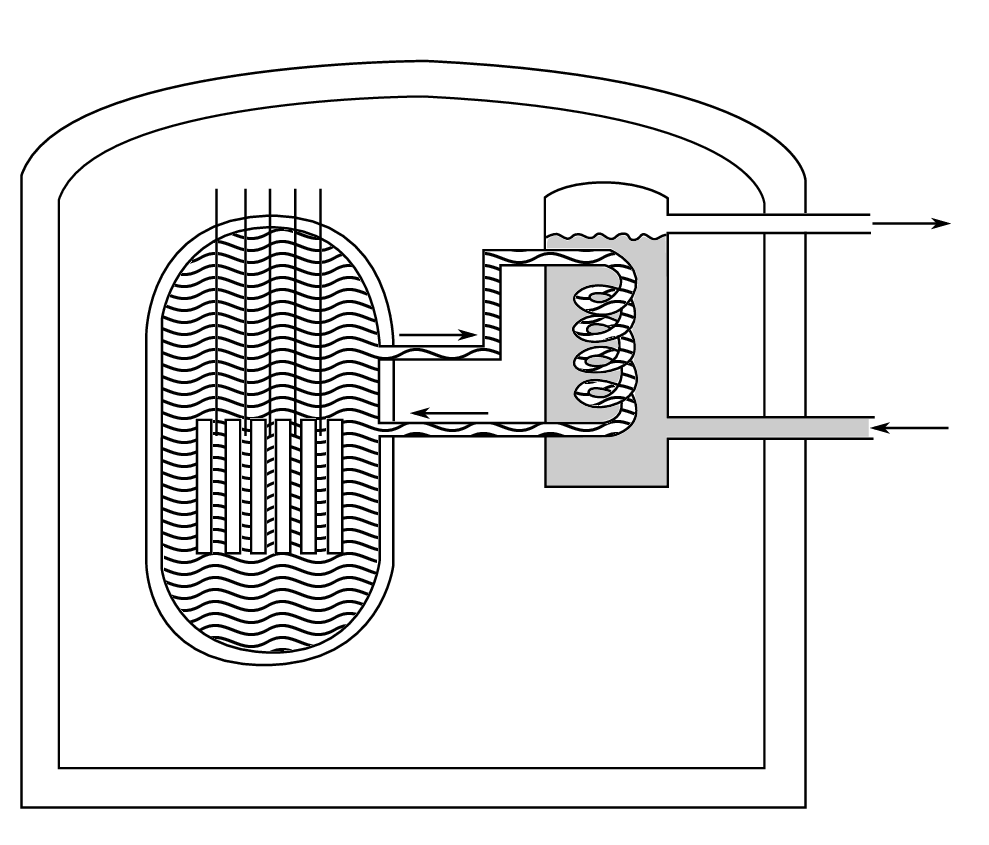
In this case, the circulating water extracts the heat used for steam generation and acts as a moderator for the neutrons. The reactor is controlled with neutron-absorbing control rods, usually made of cadmium.
4.5 Exercises
Example 4.5.1.
The radius of an azimuthally symmetric distorted nuclei can be expressed as (up to second order)
where are the Legendre polynomials. Show that in order to preserve nuclear volume and the centre of mass position, and .
Solution.
This calculation was first performed by Bohr and Wheeler.
The Legendre polynomials are , , . Since is an odd function, and and even, it must be that otherwise the centre of mass will change.
Now let us perform the volume integral
Note the integral over has been performed before since the upper integration limit of is determined by . We now have
where . Expanding out and keeping terms to (since we are interested in small distortions),
We can now use the orthogonality condition of the Legendre polynomials
We therefore have
For volume to be conserved
Again we expand out and keep terms
The solution to this quadratic equation is
and using the binomial expansion we find .
Example 4.5.2.
For an ellipsoid with semi-major axis and semi-minor axis , with volume , the deviation from a sphere with radius can be characterised by , where
| (4.9) | |||||
| (4.10) |
Show that the change in the surface energy contribution to the binding energy is modified by
| (4.11) |
and the change in the Coulomb term is
| (4.12) |
Hence calculate the difference in binding energy between the ellipsoid and sphere.
